MyHealth@Eu Laboratory Report
9.1.1 - trial-use
150
MyHealth@Eu Laboratory Report
9.1.1 - trial-use
150
This page is part of the MyHealth@Eu Laboratory Report (v9.1.1: Wave 9) based on FHIR (HL7® FHIR® Standard) R4. This is the current published version in its permanent home (it will always be available at this URL). For a full list of available versions, see the Directory of published versions
| Official URL: http://fhir.ehdsi.eu/laboratory/ImplementationGuide/myhealth.eu.fhir.laboratory | Version: 9.1.1 | |||
| Active as of 2026-05-05 | Computable Name: MyHealthEuLaboratoryIg | |||
Specify a set of rules to be applied to HL7 FHIR to define how to represent a Laboratory Report for the European cross-borders exchange, coherently with the European eHN Guidelines (see the European eHealth - Key documents ).
This Implementation Guide applies to laboratory reports within the core fields of in-vitro diagnostics (e.g. clinical biochemistry, haematology, microbiology, …), while leaving out some specialised laboratory domains like histopathology or medical genetics.
This guide is derived for the HL7 Europe Laboratory Report HL7 FHIR IG fulfilling the MyHealth@EU Functional Requirements (see the MyHealth@EU Laboratory Models Overview page)
The Use Case originates from the Change Proposal produced by the X-eHealth project/MyHealth@EU New Use Case Workgroup, and the details are available in the corresponding Change Proposal: CP-eHealthDSI-073: Implement the new service Laboratory Result Report (corresponding to the Wave 8 of MyHealth@EU).
The corresponding eHN Guidelines: eHN Laboratory Result Guidelines (release 1) are available here
The solution adopted by this guide - and detailed in the HL7 EU Laboratory Report Design choices page - balances the business requirement of Laboratory Report as legally signable document (i.e. as a HL7 FHIR document), with the expectation to get Lab Report by searching per DiagnosticReport. All this, taking into account the R5 DiagnosticReport design pattern where the DiagnosticReport - Composition relationship is directed from the DiagnosticReport to the Composition resource.
This is done by supporting both perspectives (see figure below) requiring the document bundle ( BundleLabReportMyHealthEu ) to always include a DiagnosticReport ( DiagnosticReportLabMyHealthEu ) and enabling the pre-adoption of the R5 rules for the inclusion of entries in the Document Bundle.
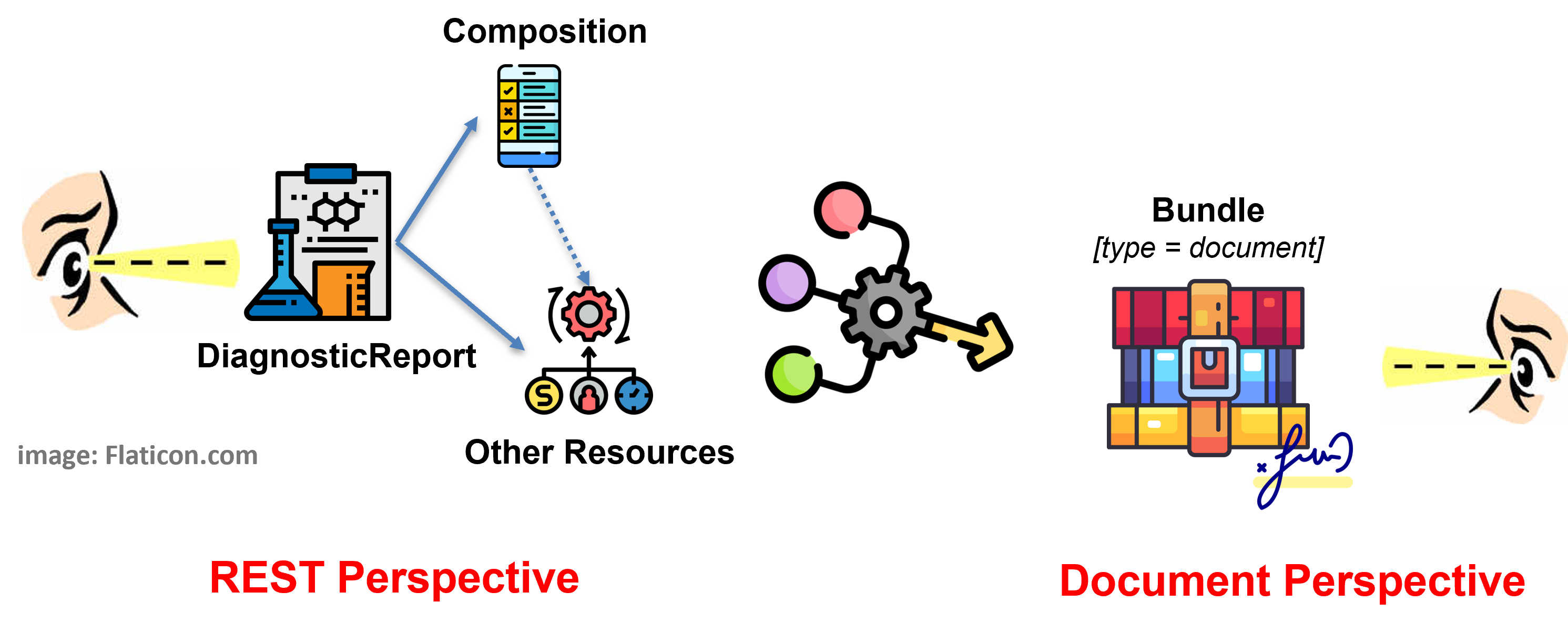
Figure 1 - Overview of the report design approach
The following diagrams provide a browseable overview of the profiles specified by this guide (not all the relationships have been reported).
The first highlights the most relevant relationships starting from the DiagnosticReport ( DiagnosticReportLabMyHealthEu ) resource (REST Perspective).
Figure 2 - Overview of the profiles relationships
The second the profiles included in the document bundle ( BundleLabReportMyHealthEu ) (Document Perspective).
Figure 3 - Overview of the document structure